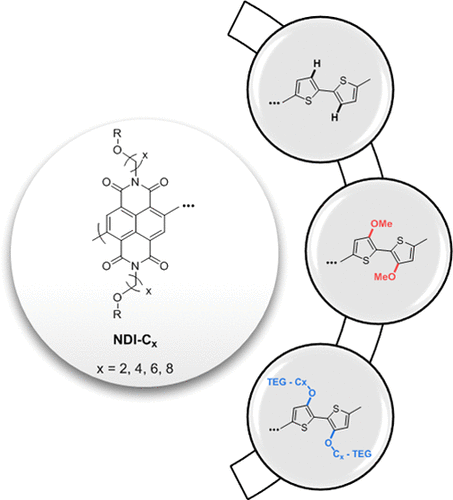
n-Type (electron transporting) polymers can make suitable interfaces to transduce biological events that involve the generation of electrons. However, n-type polymers that are stable when electrochemically doped in aqueous media are relatively scarce, and the performance of the existing ones lags behind their p-type (hole conducting) counterparts. Here, we report a new family of donor–acceptor-type polymers based on a naphthalene-1,4,5,8-tetracarboxylic-diimide-bi-thiophene (NDI-T2) backbone where the NDI unit always bears an ethylene glycol (EG) side chain. We study how small variations in the side chains tethered to the acceptor as well as the donor unit affect the performance of the polymer films in the state-of-the-art bioelectronic device, the organic electrochemical transistor (OECT). First, we find that substitution of the T2 core with an electron-withdrawing group (i.e., methoxy) or an EG side chain leads to ambipolar charge transport properties and causes significant changes in film microstructure, which overall impairs the n-type OECT performance. We thus show that the best n-type OECT performer is the polymer that has no substitution on the T2 unit. Next, we evaluate the distance of the oxygen from the NDI unit as a design parameter by varying the length of the carbon spacer placed between the EG unit and the backbone. We find that the distance of the EG from the backbone affects the film order and crystallinity, and thus, the electron mobility. Consequently, our work reports the best-performing NDI-T2-based n-type OECT material to date, i.e., the polymer without the T2 substitution and bearing a six-carbon spacer between the EG and the NDI units. Our work provides new guidelines for the side-chain engineering of n-type polymers for OECTs and insights on the structure–performance relationships for mixed ionic–electronic conductors, crucial for devices where the film operates at the aqueous electrolyte interface.